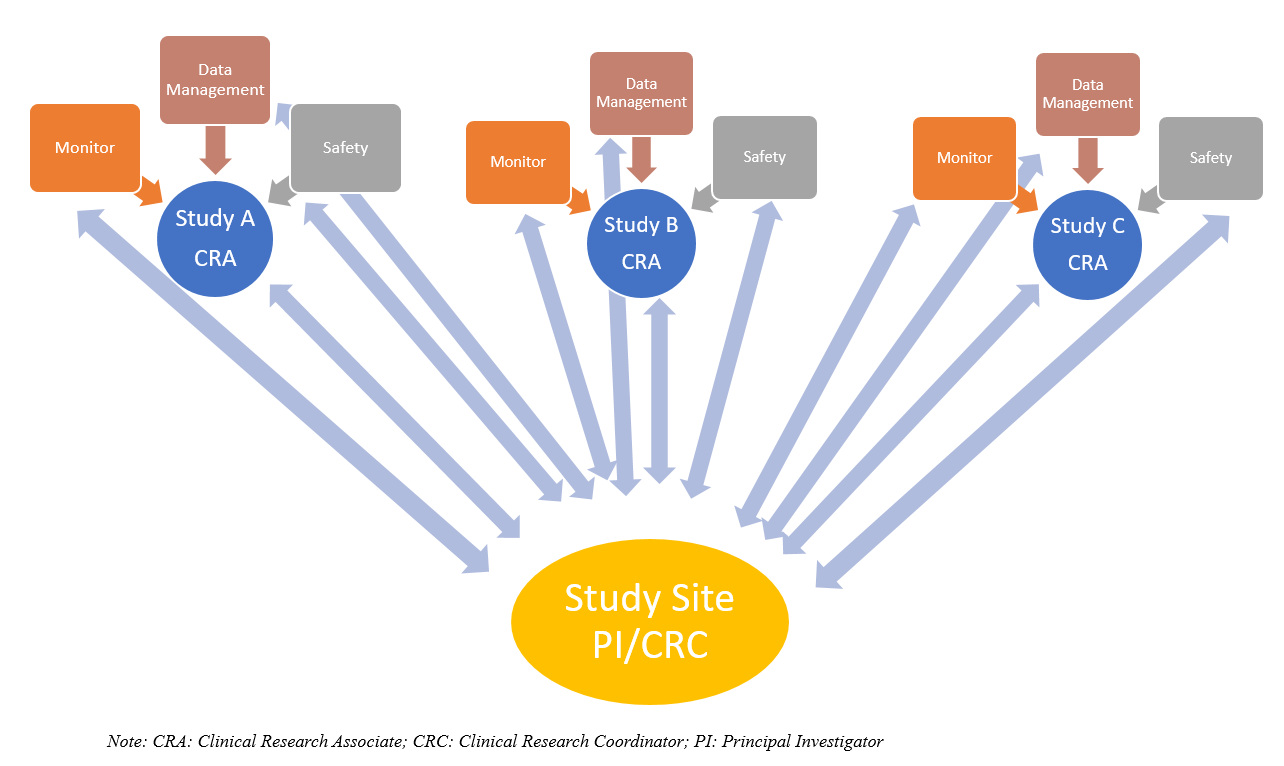
Sponsor-Site Communication in Device Trials: Evolution of a Dedicated Field Clinical Organization Throughout Study Execution - ACRP

WHEN, WHY AND HOW SPONSOR, CONTRACT RESEARCH ORGANISATION (CROs) AND RESEARCH SITES WORK TOGETHER: – your pharmacy guide

The Good Clinical Practice (GCP) and the responsibilities of pharma sponsors - Avantyo article in Viata Medicala magazine · News · Avantyo